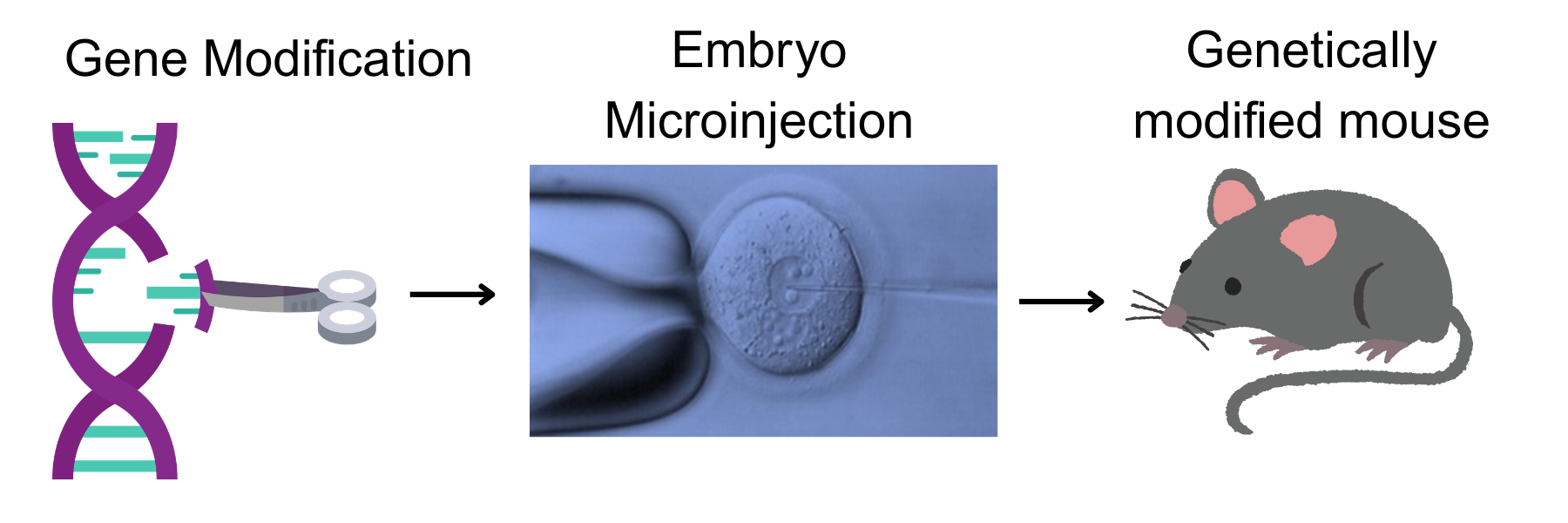
The February 2026 issue of the Research Resource Nexus highlights the Innovative Technologies Development (ITD) Core and Mouse Embryo Services (MES) Core, which work together to provide investigators with the knowledge and experience to produce new and custom genetically engineered mouse models.
Innovative Technologies Development Core
ITD Core director, Sebastien Gingras, has 25 years of experience in creating and using complex genetically engineered mouse models. Gingras collaborates with investigators to design the gene targeting strategy best suited for their project. Projects begin with in-depth experimental design consultation to ensure allele architecture, editing strategy and downstream genotyping approaches are optimized before embryo work begins. From allele design through founder identification and genotyping strategy, the cores provide end-to-end design, reagent evaluation, embryo manipulation and validation support, removing the logistical and technical burden of mouse model generation so investigators can focus on the biological questions rather than the engineering process. Using CRISPR/Cas9 technology or classical transgenesis, ITD Core has extensive experience designing and implementing complex alleles, including but not limited to:
- knockout;
- targeted mutation (point mutation and SNP);
- conditional knockout;
- knock-in and conditional knock-in in the ROSA26 locus;
- gene-specific knock-in (reporter mice with fluorescent protein and gene-specific recombinase, e.g., Cre, Flpo); and
- classical random insertion of transgene.
Mouse Embryo Services Core
MES Core performs embryo microinjections with the modified DNA or RNA generated by ITD Core and supports a variety of projects using several types of mouse strains, including the widely used C57BL/6, and others, such as BALB/c, 129S1/SvlmJ, MRL and CBA. The core provides a comprehensive range of services to help investigators generate, recover, preserve and maintain genetically engineered mouse lines, safeguarding valuable lines and ensuring long-term reproducibility of research programs. Services include:
- rederivation, or recovery, of mouse strains from frozen embryos or sperm through in vitro fertilization or intracytoplasmic sperm injection;
- cryopreservation of sperm and embryos, including long-term storage;
- sperm motility testing and mice pathogen testing;
- import, export and relocation of cryopreserved materials; and
- consultation on mouse colony maintenance and breeding.
MES Core director, Chunming Bi, has 25 years of experience in mouse reproductive biology and mouse embryo micromanipulation, including 14 years in gene targeting mouse generation. Together, ITD and MES Cores have produced more than 300 mouse strains since their establishment in 2014, supporting numerous successful grant applications and publications. To initiate a project or for more information, contact ITD Core by emailing Gingras at sgingras@pitt.edu, or visit the ITD Core website. For MES Core, email Bi at chunming@pitt.edu, or visit the MES Core website or MES iLab site.
Testimonials
Adam Straub, professor and vice chair for research in the Department of Pharmacology and Chemical Biology, School of Medicine, studies how redox signaling and genetic variation contribute to heart and blood vessel function and how this affects conditions, such as cardiovascular disease, stroke, hypertension and sickle cell disease. Because this research involves complex interactions that cannot be fully studied in cells or patient data alone, genetically engineered mouse models generated by ITD and MES Cores have been essential to Straub’s research. His long-standing collaboration with the cores has led to the development of numerous gene knockout models, supporting more than $10 million in funded grants that Straub notes would not have been possible without these shared resources. Describing the impact of ITD and MES Cores, Straub shared, “Sebastien is fantastic in the design of new mouse models and has been very successful in creating models not just for our work, but for the University of Pittsburgh.” He also noted how the cryopreservation and rederivation of his mouse lines have enabled collaborations with investigators both within and outside the University. Straub emphasized how critical mouse models are for understanding disease causality, exploring complex cardiovascular physiology and examining how genetics influence drug response, ultimately advancing precision medicine and translation to human disease.
Arohan Subramanya, associate professor of medicine and of cell biology, School of Medicine, studies how the kidneys sense and respond to changes in potassium levels to regulate electrolyte balance and blood pressure, with a focus on a group of enzymes called WNK kinases. With the support of ITD and MES Cores, a mouse model with a mutation in the WNK1 gene was generated to demonstrate how specialized structures, called WNK bodies, help the kidneys conserve potassium under stress. This in vivo model provided key evidence connecting potassium sensing to salt handling and blood pressure regulation, insights that would have not been possible using traditional knockout approaches. The findings, published in the Journal of Clinical Investigation, helped Subramanya and colleagues close an important gap in the field of kidney physiology and could have implications for hypertension, cardiovascular disease and stroke.